Testing One
I have many friends who take an interest in my pots of goo and occasionally they send me things. One such friend returning from Essaouira sent me pigment she had been sold as ‘shellfish purple’. Historically, the Moroccan coast was an area much involved in the making of this fabled dye (also known as Imperial Purple and Tyrian Purple) but I expressed doubt that what she had sent was ‘the real thing’ because it is fabulously expensive to produce even a small quantity. Just 5 grams costs around £450.00. But I thought it would be fun to try dyeing with it.

Green grains and finished colour on silk
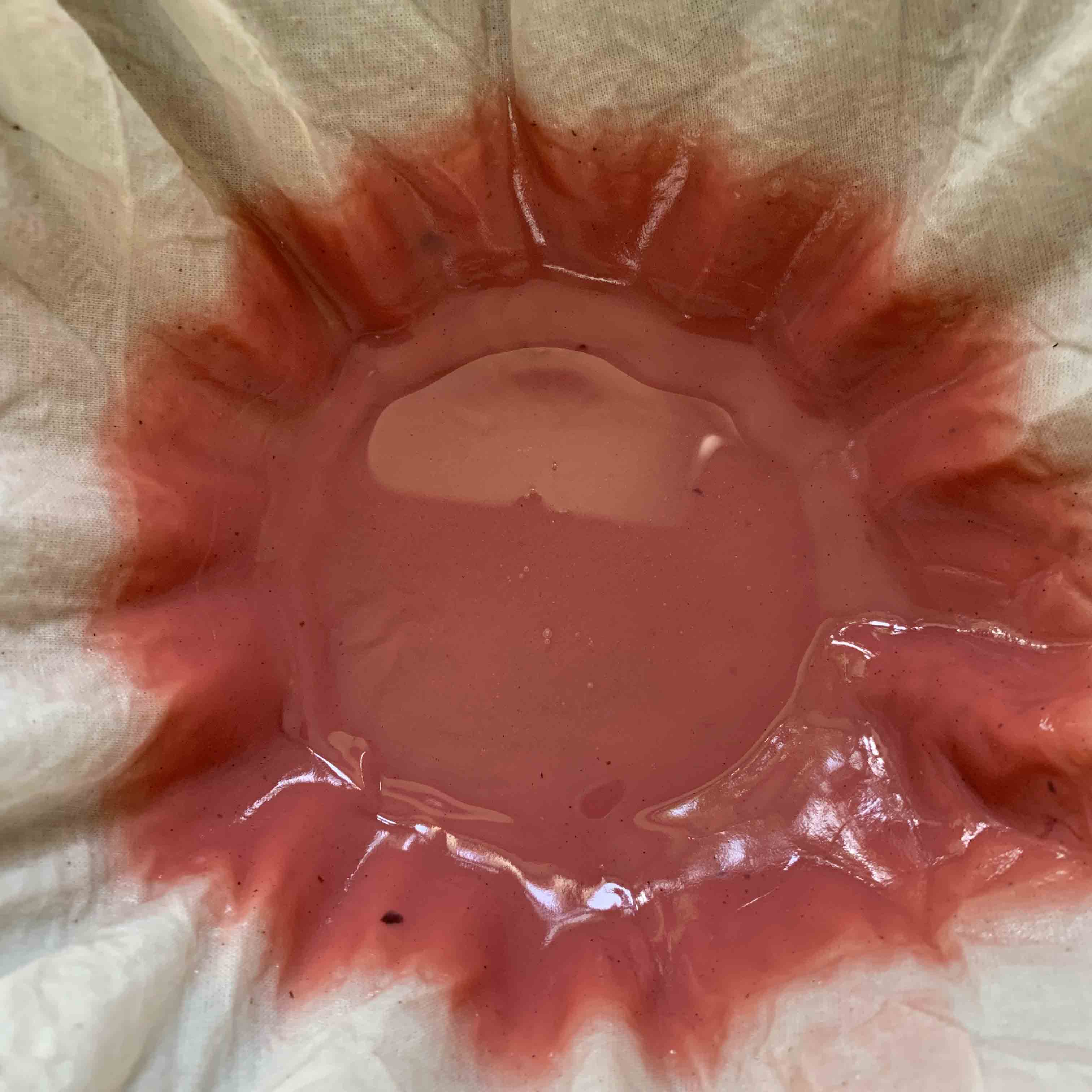
I took advice from a specialist colleague, Professor Zvi Koren of the Edelstein Center for the Analysis of Ancient Artefacts (link below). He advised that shellfish pigment is not water-soluble, so that by adding just water I would not get a true solution: I’d get a reddish coloured mixture with the pigments dispersed but not dissolved. I added water and the grains went bright pink. Very bright indeed, as you can see below.

The colour of the grains in solution: testing pH

Reduced liquid with lumps of dye matter
Following my colleague’s advice, I reduced grains in a hydrosulfite / dithionite bath in an alkaline solution, at about 60 C. The liquid went completely clear, with the dye matter gathered in lumps. This didn’t look right at all for shellfish purple, which should change to a greenish colour (as with indigo). Nevertheless, I dipped silk into the clear mixture and it came out a bright pink – which does not wash out. So it’s certainly a dye, but certainly not shellfish purple.
Two other pointers to its not being shellfish purple: the Essaouira grains are green, and they shouldn’t be. There’s no snail pigment that colour, according to my colleague. It’s usually dark, blackish, brownish, purplish or violetish, but never green. And on top of that, the grains should have a yukky fishy smell. The Essaouira grains smelled vaguely of incense.
So this was a fascinating experiment, a story echoing many historical tales of dyes that were not as they claimed.
My thanks to Professor Zvi Koren of the Edelstein Center for the Analysis of Ancient Artifacts for his input and advice on testing the Essaouira grains.
Friends: please don’t stop sending me interesting things. But as an H&S caveat, pigments and grains bought in faraway places may be toxic or otherwise harmful so you need to be aware of handling and storing them. They are not necessarily what they say on the tin.
Testing the Other
At a recent course at West Dean my Old Geyser of a fabric steamer developed a problem. The thirty-year old steamer assembly consists of a standard water boiler (the sort to be found in every village hall), a custom-made stainless steel chamber, lid, and perforated base plate. Silks are rolled in paper and stacked upright inside the steel chamber. Water is heated in the boiler, the steam circulates and the combination of heat and damp sets the dyes. The water isn’t held at a constant boil but needs to come up to the boil – and hold it – every two or three minutes, for around two-and-a-half hours.
Only, at West Dean, it didn’t. The boil was less frequent than usual, and was held for shorter periods. I was concerned that dyes were insufficiently fixed and suspected a problem with the thermostat. (I should add that West Dean supply a professional Uhlig steamer, but I have always used mine, which holds more silk).
Back home, phone calls revealed that a new ‘simmerstat’ is what I required. But during the time since I bought the boiler, Brussels has dictated that EU citizens are insufficiently responsible to handle dangerous pieces of equipment that boil water. (Those in favour of Brexit might enjoy the link at the bottom of the page). A catering boiler will no longer come to a full, constant boil. My new simmerstat was fitted by the technical department, but the gaps between boils seemed longer than I remembered…. or was I just being twitchy?
I then discovered that a secondhand Uhlig steamer was on sale, owned by an ex-student. I couldn’t believe this piece of luck – and bought it. It is a solid, stable and well designed piece of equipment, although as with the West Dean one, it does not hold as much yardage as Old Geyser. In the Uhlig I tested several pieces of silk, including three blues which have a tendency to run if steamed sufficiently. No run-off.

Some runoff may be expected in initial rinses after steaming if heavily concentrated dye is used. Thereafter the water should run clear

Samples of identically dyed silks steamed in two steamers to compare colour and runoff
I tested identical blues in the mended Old Geyser. It now appears to be working well too – so I now have two working steamers. No recycling tip for Old Geyser: he threw a steamy party.
LINKS
Where to buy shellfish purple in 2016? Here
Brexit? Pulling the plug on high speed kettles here
The Edelstein Center for the Analysis of Ancient Artifacts here